Melissa Cody, APRN Pulmonary/Critical Care
We are all required to interpret an arterial blood gas at some point in our career. If you’re me you will certainly be looking at them all day; everyday. As an ACNP who works with a Pulmonary / Critical Care team ABGs are also a huge part of my daily patient management. That being said, it is necessary to have a basic understanding of these concepts regardless of your specialty.
Here is a great example: You’re working with a hospitalist team and your patient just came up from the ER with a UTI and some mild hypotension. The ABG shows a pH 7.028 pCO2 28 pO2 128 HCO3 5 BE -16. Your patient has a metabolic acidosis. How did you determine that?
pH – The Acid-Base Determinant
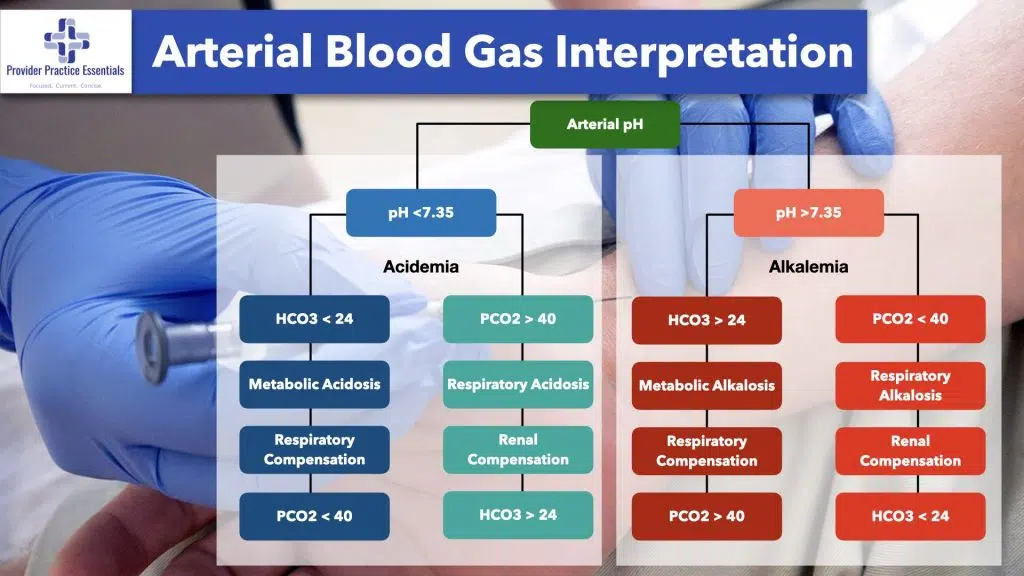
Let’s start with pH – the first value reported in an ABG. The human body does all that it can to maintain a pH in the range of 7.35 – 7.45. Anything below 7.35 is acidotic — the patient either has too much acid built up or too little base (Bicarb). We use primarily the kidneys and lungs to help control this balance. The lungs are certainly our quickest means of controlling the pH. When we are becoming acidotic (pH<7.35) our brain tells us to speed up respirations in order to blow off additional carbon dioxide. The kidneys are a bit slower to help out and take several days to adjust the amount of acid/base excretion.
Carbon Dioxide (PCO2) and Bicarbonate
We can move to the balance of carbon dioxide and bicarb next. As mentioned above these will be the major players in affecting the pH (Acid-Base) balance reported in an ABG. pCO2 stands for the partial pressure of carbon dioxide (acid). The normal range of pCO2 is 35 – 45 mmHg (with some variation for chronic CO2 retainers — think COPD). The HCO3 (bicarb) on an ABG is a calculated value. Therefore, the reported serum bicarb value should be factored In to the interpretation. That being said, a normal range for HCO3 is 22-26 mEq/L. Bicarb is your base.
Let’s pull together what we’ve learned so far about interpreting an arterial blood gas. pCO2 is an acid and HCO3 is a base. If the pH is less than 7.35 than you know your patient is acidotic. Why? That is when we move to our acids/bases. If you have an elevated pCO2 and normal HCO3 → then you have a respiratory acidosis on your hands. If you have a normal pCO2 and low HCO3 then you have a metabolic acidosis. SO REMEMBER: As your pCO2 will be inverse to your pH → as your pCO2 rises your pH will fall. Conversely your pH follows your HCO3. As your HCO3 falls so will your pH.
Oxygen (PO2)
Lets move on to pO2. This is the partial pressure of oxygen. The normal range is 75-100mmHg. Oxygenation is measured by the the PO2. It is often used to calculate a P/F ratio in ARDS. It can also help to determine respiratory failure in patients with COVID. This ratio takes pO2/FiO2 to determine the level of respiratory failure. Therefore it is used as a diagnostic tool in the clinical management of these patients. For our purposes — if it’s low, they need more oxygen support.

Base Excess
The base excess (BE) determines the degree of metabolic derangement. Likewise, a normal BE should be zero but -3 to +3 is generally considered acceptable. This part of the arterial blood gas also helps us determine the amount of base needed to reach a normal pH. If there is a base deficit, it will certainly show up as a negative base excess (negative number). So in high BE is the same as a high HCO3 for purposes of ABG basics.
That is basic ABG interpretation! Let’s go over a few examples for practice and I have also included some helpful diagrams below.
ABG Examples
#1: A 63 y/o F presents to your ER with h/o 2ppd tobacco use and 2 days or progressively worsening SOB. ABG: pH 7.23 pCO2 81 pO2 93 HCO3 26 BE 3
Answer: Respiratory Acidosis
Rationale: Your pH is low (acidosis), pCO2 high (respiratory) and serum bicarb is normal
#2: A 32 y/o F poorly controlled diabetic is admitted to your ICU with DKA. ABG: pH 7.013 pCO2 22 pO2 145 HCO3 7 BE -12.
Answer: Metabolic Acidosis
Rationale: pH is low (acidosis), pCO2 is low (alkalosis but not enough to compensate), HCO3 low (acidosis)
#3: A 76 y/o chronic COPD patient presents to your urgent care with c/o cough and increased SOB. ABG: pH 7.37 pCO2 62 pO2 95 HCO3 32
Answer: Compensated respiratory acidosis
Rationale: pH (normal), pCO2 elevated (acidosis), HCO3 elevated (alkalosis)
#4: A 21 y/o is brought into the ER with 4 days of dyspnea, fatigue and fever.
ABG: pH 7.4 pCO2 35 pO2 40 HCO3 24 BE 2
Answer: Hypoxia, normal acid base
Rationale: pH, pCO2 and HCO2 are all within normal ranges. pO2 is low.
Additional information